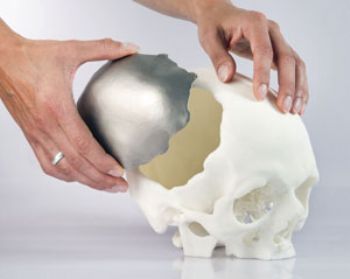
Slovakian-based CEIT Biomedical Engineering has started to use additive manufacturing technology to give doctors precise bone replacement material for cranial implants.
The implant must fit precisely to ensure the long-term success of a surgical operation and allow the patient to live without discomfort. CEIT Biomedical Engineering was created by the Technical University of Košiceto promote additive manufacturing in the medical sector (
www.ceit-ke.sk).
Professor Radovan Hudak, managing director, said: “We wanted to explore the potential of additive manufacturing for implants and at the same time develop solutions that were both helpful to patients and economical.
Precision, reproducibility and surface quality were all high on the list of requirements, along with a production process that was as free as possible from production errors. The main aim was then to acquire state certification for cranial, jaw, and facial bone implants.”
The CEIT team took a year to explore the market, eventually choosing EOS as its technology provider and EOS titanium alloys as its starting material. The first implant it produced was a piece of replacement cranium about 15cm across and 1.5mm thick, made from titanium Ti-6Al-4V and weighing 63g. A notable feature was the hollow lattice structure, which promotes the growth of bone tissue and allows the integration of micro-sensors.
This type of implant benefits from higher precision and a lower error rate in production, resulting in fewer side-effects and quicker recovery for the patient, plus less evidence of the procedure once the wound has healed. The process has now been certified, resulting in official EU-wide approval.